1 juillet 2020 – Policy Brief
Strategy to react to substantial increases in the numbers of SARS-CoV-2 infections in Switzerland
Download Policy Brief PDF
Cette Policy Brief n’a pas été traduite en français.
Dans la version actuelle, les erreurs de frappe et les hyperliens ont été corrigés.
La première version du 8 juin 2020
Summary of request/problem
FOPH provided the ncs-tf with ten specific questions about how to respond to substantial increases in the numbers of SARS-CoV-2 infections in Switzerland. The ncs-tf addressed these questions and also provided a general perspective on the strategy that it recommends.
Main text
This document presents a surveillance-response strategy that covers different scenarios including substantial increase in case numbers of SARS-CoV-2 infection in Switzerland. Section A describes the overall approach of a surveillance-response strategy with four key considerations:
1) introduce the response early,
2) implement interventions at the appropriate geographic scale,
3) implement specific measures in the right sequence, and
4) define the responsibilities and financial issues.
Section B applies the surveillance-response approach to ten questions from the FOPH about different aspects of a second wave.
A) General considerations
1) Timing of the surveillance-response strategy
2) Surveillance and responses at different spatial scales
A key idea of the surveillance-response approach is that interventions occur at the appropriate spatial scale. Interventions should only be implemented in regions where they are needed. On the other hand, interventions have to cover the whole region where an outbreak occurs, to prevent flaring-up after the termination of the intervention.
Choosing the spatial scale for interventions is not only based on epidemiological considerations. While life and work situations are not confined by political boundaries, decision-making processes and budgets are. Cantons and communes must, therefore, be important actors. They are the only entities able to take legitimate restrictive measures and spend public money. Cantons and communes should be encouraged to cooperate within regions of closely interconnected cantons and communes, as they are already used to do.
A spatial scale that makes sense from an epidemiological, as well as a political perspective, is that of the “Sentinella regions”. The six Sentinella regions are, for example, used by FOPH for influenza. We propose that the Sentinella regions should play an important role in the surveillance of case numbers and the implementation of additional measures.
We need to complement the approach based on the Sentinella regions by considering both smaller and larger spatial scales for surveillance and responses. We expect that many outbreaks will affect small areas and might be confined to, for example, a retirement home. In such cases, interventions on this small scale are appropriate. On the other hand, as we discuss in more depth in an upcoming policy brief, a substantial part of the Swiss population lives in border regions that are connected with neighboring countries: it is vital then that these neighboring countries be involved in a coordinated response.
Difficulties may arise when implementing a spatially targeted surveillance-response approach. Recent research shows that measures that are targeted to specific regions often affect areas beyond these regions (source). This is a consequence of mobility across the boundaries of individual regions as well as so-called spill-over effects: interventions in a region alter the behavior of individuals in places where these interventions do not apply. It will be essential to closely monitor not only the spatial dynamics of infections, but also the behavior of the population as well as the economic consequences of altered behavior in different regions.
3) Sequence of interventions
The sequence in which additional interventions should be introduced in case of an increase in the number of cases is an important question. We assume that the basic prevention measures, including social distancing, hygiene and selective mask wearing, will remain in place for some time: these measures are an essential pillar of Switzerland’s strategy to control SARS-CoV-2 until an effective vaccine or treatment becomes available.
If the number of new infections increases substantially in a certain region, additional measures should be considered. We provide a provisional list of such measures and the sequence in which they could be most effectively applied. Measures listed first are less disruptive and should be implemented first, while subsequent, more severe measures are adopted later.
- Intensified and targeted communication. The behavior of individuals has a strong influence on the rate of transmission, and focused communication campaigns can be used as a fast response to increasing case numbers. Communication campaigns need to be targeted for specific regions and situations. This includes e.g. situations at the border with cross-border commuters.
- Promotion and enforcement of mask wearing. WHO has established that medical masks can reduce transmission by up to 80% (source). We recommend medical masks i) for staff working with any type of patients in healthcare facilities; ii) people aged 60 years or over, as well as those with underlying conditions, in situations where physical distancing is not possible; iii) masks for the general population where there is widespread transmission potential and physical distancing is difficult, including on public transport, in shops or in other confined or crowded environments.
- Measures to isolate and protect groups at high risk of adverse outcomes from a COVID-19 infection.
- Banning of events with large numbers of participants.
- Temporary closure of settings with high potential of transmission.
4) Surveillance and response - responsibilities and financial considerations
An effective surveillance and response plan requires clarity about who is in charge of deciding on and implementing measures. Are cantons (or cities) allowed to adopt certain measures? How far can they go? Are there national thresholds and limits? Or is the easing of measures binding for all actors until the Federal Council decides otherwise? Lack of clarity on these issues will impede timely reactions.
Financial matters must also be clarified in order to be able to respond in a rapid and appropriate manner. The general rule that the cantons bear the cost of implementation should be the guiding principle. As there is a constitutional principle saying, “whoever pays, commands”, autonomy and cantonal diversity depend on autonomous financial sources. Federal finances, as tempting as they may be in times of crisis, limit the cantonal room for manoeuvre and run the risk of uniformity at the cost of diversity. The Confederation must contribute to bearing the economic burden – but it should do so within its competencies (health insurance, unemployment insurance, etc.) and abstain from using emergency powers to interfere with cantonal matters and to govern by the purse.
Of course, cantons are bound to all federal laws and ordinances are binding on the cantons (such as the Epidemics Act and Covid-19 ordinances) and federal authorities have monitoring and supervisory powers.
B) Specific questions
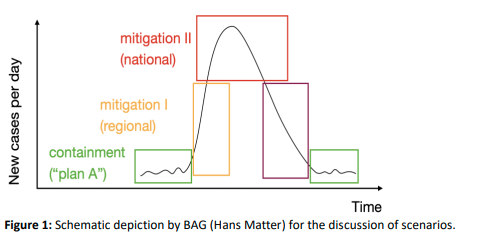
1) What are possible scenarios for the development of the COVID-19 epidemic in Switzerland?
Different scenarios are conceivable and require different sets of responses, including responses at different spatial scales. We consider four scenarios:
- Isolated clusters that can be locally contained by means of the cantonal capacities of surveillance and response (TTIQ). Such small and isolated local outbreaks will not challenge cantonal capacities for contact tracing, testing, isolation, quarantine and hospitalization. A regional or national estimate of the reproduction number Re may not be appropriate to monitor this situation, because low case numbers lead to large uncertainties in the estimate of Re. A useful indicator of the effectiveness of contact tracing is the fraction of cases that can be epidemiologically linked to other cases (“traceability”). Another indicator is the time span between the onset of symptoms and the isolation/quarantine of index cases and their contacts. The smaller this time span the more effective contact tracing will be in reducing transmission. High traceability and fast isolation of index cases and their contacts indicate that the containment effort is successful.
- Larger or multiple regional clusters that require coordinated action between several cantons (this will often be the “Sentinella regions” introduced above). This requires efficient intercantonal exchange on TTIQ. Again, high traceability of cases is an important indicator of the effectiveness of the containment measures. In this scenario we do not foresee that the maximal capacities of TTIQ and hospitalization will be overwhelmed.
- Larger regional outbreaks that cannot be contained by TTIQ and lead to sustained exponential growth of the epidemic (within and even beyond a “Sentinella region”). Key indicators in this case are an effective reproduction number Re larger than 1 and a decline in traceability. Such outbreaks will require the fast implementation of additional regional measures beyond TTIQ. A delayed response carries significant risks as mobility may rapidly lead to secondary outbreaks in other regions. Such outbreaks may overwhelm local or regional capacities for contact tracing and challenge hospitalization capacities and thus require coordination across regions.
- Multiregional outbreaks with sustained exponential growth (such as observed in the first wave). Key indicators are again a Re > 1 and low traceability. Such outbreaks will eventually overwhelm the capacity of the healthcare system, if no appropriate actions are taken on a multiregional or even national level. This scenario will only arise if control fails under scenarios 1-3 and will require the reintroduction of costly general measures. Failure to control this scenario will lead to even higher costs in terms of health and economy,and should therefore be avoided by all means. Given that the first wave resulted in over 1500 deaths and a seroprevalence of a few percent only, an uncontrolled second wave could lead to many more additional deaths.
Scenarios 1 and 2 correspond to “containment” in Figure 1, with scenario 2 requiring regional coordination for containment. Scenario 3 corresponds to “mitigation I (regional)”, characterised by epidemic growth and requiring regional action and coordination. Scenario 4 corresponds to “mitigation II (national)”, requiring multiregional or national coordination and action.
The scenarios are directly relevant for planning and managing the capacity of the healthcare system and specifically ICU capacity. Scenario 2 requires coordination in surveillance and response (specifically TTIQ), but likely limited coordination regarding hospitalization. We recommend that capacity for intercantonal TTIQ be urgently developed. In scenario 3 or “mitigation I” it is essential to prevent exceeding local or regional capacities by coordinated management and transfer of patients to regions with free capacity. In scenario 4 or “mitigation II”, rapid coordination is essential to make full use of the ICU (intensive care unit) capacity across Switzerland. The ICU capacity in Switzerland could be increased to 1300 beds, and we ecommend that hospitals are prepared for a swift increase to such capacity. We also propose to set up a national coordination group for ICU beds. National coordination is vital to avoid situations where unaffected regions are reluctant to share their ICU resources out of concerns that they might subsequently need these beds themselves.
2) What are the main unknowns with respect to the development of the COVID-19 epidemic in Switzerland? For example, what are unknowns with respect to new disease syndromes, new insights about risk groups etc.
We distinguish between scientific and operational unknowns. The former will be shaped by the progress made on the science of COVID-19, which the task force is closely monitoring. The latter are primarily linked to implementation issues at various levels.
Scientific unknowns:
- Immunity: The nature and duration of immunity following SARS-CoV-2 infection remain unknown. This has implications for understanding the value of serological testing at the individual level, the interpretation of results of population-based testing, and the likelihood of successful vaccine development and evaluation.
- New disease syndromes: The occurrence of new syndromes seems unlikely. The clinical picture in adults has so far been uniform across very different locations worldwide. Information regarding medical care, as stated in the policy brief about the care of the elderly population, has to be in place from the beginning with information on the serious prognosis in frail patients. Severe diseases in children occur very rarely. Kawasaki-like syndromes were described during the Covid-19 epidemic. The frequency is estimated to be at 1 in 1’000 children. Mostly the prognosis was good and full recovery reached.
- Risk groups: We have a reasonably good understanding of the risk factors for severe COVID-19. These predictors are described in our policy brief of May 1, 2020. In addition to these clinical factors, socioeconomic factors have been recognized as having a substantial effect on the probability of contracting SARS-CoV-2 and on the probability of severe outcomes of COVID-19. In several countries, disadvantaged populations have been shown to be at greater risk of infection, for example people of color in the United States, or migrant workers in Singapore.
- Effects of seasonality: Current knowledge suggests that seasonality has only a small effect on the spread of the pandemic (source). One relevant seasonal risk factor, however, is influenza. The superposition in autumn/winter of an increase in COVID-19 cases and the flu outbreak could stretch the hospital capacity. A policy brief on this topic is in preparation.
- Role of superspreading: The role of superspreading is still actively debated. This debate is related to the discussion of the role of aerosol transmission in the spread of COVID-19. We expect that knowledge will accrue on the quantitative contribution of both superspreading events and aerosolized transmission. A better understanding of what type of events lead to superspreading will allow targeted prevention measures.
- Socioeconomic factors: We do not yet have a good understanding of which socioeconomic factors increase the risk of infection and transmission. In other countries, workers in precarious employment situations continued to work after developing symptoms because they could not afford to stay at home or feared losing their jobs. Improving our understanding of what socioeconomic factors may increase the risk of infection in Switzerland will help to target measures in the future.
Operational unknowns:
- TTIQ: The level of coordination of TTIQ in the different cantons is still unknown. Moreover, it is unclear what minimal essential data will be collected by all contact tracers in all cantons and whether/how these data will be exchanged and shared between cantons, and with the FOPH and the Science Task Force.
- Coordination of the response: Effective intercantonal collaboration with well-defined roles and responsibilities under the umbrella of the national strategy and guidelines is yet to be established. Substantial coordination efforts will be required to ensure the success of this measure.
- International context: The level and format of inter-country collaboration for cross-border issues, including cross-border TTIQ, remain a very important unknown in the future control of this pandemic.
- Testing: While the current capacity of diagnostic PCR tests is now reasonably well established, the role and capacity of alternative uncertified tests is not yet clear. Currently, there is capacity for around 20,000 PCR tests per day, which could be further increased to 25’000 per day. An additional reserve of 1 Million diagnostic tests is guaranteed by a Thermo Fischer platform. NGS (next-generation sequencing) methods could substantially increase this capacity, but are not certified. Such methods could be used for monitoring, but capacity and logistics regarding sampling and handling remain unclear.
3) What can we say about the expected dynamics of a potential second increase in case numbers? Can we make statements about the time point, size, and duration? (expected rate of increase, the expected peak height, the expected rate of decline and the expected spatial expansion?
FOPH and Science Task Force agree on the goal of preventing a second wave of cases. To achieve this, we need a maximally efficient, well-coordinated and targeted strategy of surveillance and response. We also need a continued participation of the general population in the adoption of preventive measures.
In case these measures should not be sufficiently effective to prevent a second wave, they are expected to slow the increase substantially in comparison to the initial increase in the first wave. Given substantial efforts in monitoring and fast notification of cases, we expect to detect outbreaks when they are still locally confined (see question 1). If the surveillance and response, and in particular TTIQ, are not sufficient to contain these local outbreaks, then we will get into a phase of exponential growth that is first regional and then multiregional or even national.
The size of a secondary peak depends on the rate of increase of the number of cases and on the time point of introduction as well as the effectiveness of the additional intervention measures. A fast response is essential to avoid going from scenario 3 or “mitigation I (regional)” to scenario 4 or “mitigation II (national)” (see also part A, section 1). In the absence of such additional interventions any exponential growth will eventually overwhelm our hospital capacities, potentially leading to a larger number of deaths than in the first wave (see for example source).
4) What testing strategy do we need for Switzerland and how should we adjust the testing approach to changes in case numbers? For example, how should we change the testing strategy if case numbers increase again?
This section outlines general considerations to promote testing and also specific recommendations. It follows the viewpoints that we presented the workshop with FOPH on June 2, 2020.
The central goal of the testing strategy is to make sure that people can be tested easily, even if their symptoms are mild. A central point that should be made explicit and recognized is that an individual who seeks a test is being altruistic. The strategy therefore needs to reduce barriers to testing and inconveniences for individuals. For example, testing centers could implement methods applied by blood banks – such as calls to solidarity, thanks, and snacks – for those who attend.
Summary of our specific recommendations (see also upcoming document “Summary of meeting on testing strategy at FOPH on June 2, 2020; List of specific recommendations by the ncs-tf”):
- Promote testing to individuals and increase access to and uptake of testing. Tests need to be free for individuals, individuals need to be able to reach testing centers easily and rapidly, and testing has to be promoted through a dedicated communication campaign. Family doctors and other health care practitioners should be mobilized to perform testing, and their link to testing labs should be streamlined. Less invasive methods of sampling (e.g. saliva or gargling samples) should be favored if they are as sensitive as nasal swabs.
- Find as many index cases as possible, as quickly as possible. In addition to testing all individuals with suggestive symptoms, we strongly recommend testing of the contacts. Contacts with positive virological tests become new index cases, which allows further contacts to be detected and breaks transmission chains. Contacts identified by classic contact tracing and contacts identified through digital proximity tracing should receive virological testing on two occasions. Contacts refer to ‘close contacts’ defined by criteria of proximity (<2m for >15 mins).
- Test sentinel populations who are of epidemiological importance. Extensive testing in sentinel populations serves several purposes: it allows monitoring the epidemiology in Switzerland by following populations with elevated risks of getting infected, it allows protecting vulnerable individuals and important medical infrastructure. Hospitals and elderly homes have the required infrastructure for testing, high awareness and have already been performing widespread testing. While schools do not have the necessary infrastructure and personnel readily at hand, testing campaigns in selected schools can provide important insights into the role of children and adolescents in transmission.
- Hospital admissions: This population can alert health authorities about otherwise undetected increases in case numbers. The advantages are:
– Identification of asymptomatic or pre-symptomatic SARS-CoV-2-positive patients on admission will allow fast implementation of infection prevention and control strategies, most importantly isolation precautions, thus preventing further inter- and intra-institutional spread to patients and healthcare workers.
– Early recognition of asymptomatic patients entering healthcare facilities is of particular importance, as close contact cannot be avoided and transmission events would target a) a particularly vulnerable population (i.e. hospitalized patients) and b) healthcare workers, potentially further limiting capacities and resources of the healthcare system.
– Admission screening may assist healthcare institutions in logistical and organisational issues, such as assignment of patients to specific wards, separate workflows in operating theaters, etc., in addition to guiding the use of personal protective equipment.
– Broad screening of asymptomatic patients should be considered, if widespread transmission is suspected to have occurred within healthcare facilities. Thus, rather than testing patients and healthcare workers according to classical contact tracing, ward- or facility-wide testing may result in faster identification of further cases and thus should be considered rapidly to complement classical contact tracing.
– Preoperative screening of patients may be considered as postoperative pulmonary complications have been shown to occur in half of the patients with perioperative SARS-CoV-2 infection and may be associated with high mortality (source). - Retirement homes and nursing homes: In the first wave of SARS-CoV-2, outbreaks took place in retirement and nursing homes in Switzerland and many other countries. Elderly people are at high risk of becoming infected and of severe disease and death. Healthcare and care workers and visitors have many contacts per day in these settings, increasing the risk of transmitting infection. Testing of residents and staff in these settings can identify asymptomatic/pre-symptomatic infection and allow early intervention when cases are detected.
- Healthcare workers: This is a population at high risk of SARS-CoV-2 infection.
- Schools: Despite the evidence suggesting a low risk of transmission in young children, the role of older children is still unclear. Outbreaks in Israel and Canada suggest that transmission of SARS-CoV-2 in schools might become more common, especially as more schools open and social contacts increase. Teachers are a sentinel population in schools because adults are more likely than children to present with symptomatic infection.
- Other populations with large numbers of social contacts, e.g. sales personnel.
- Hospital admissions: This population can alert health authorities about otherwise undetected increases in case numbers. The advantages are:
- Response to cases in sentinel populations: If two or more individuals in a given setting test positively, we recommend launching a dedicated outbreak plan. This plan includes broad testing across the setting and quarantining of contacts. Unless incidence increases markedly, testing in batches may be considered for individuals beyond the immediate contacts of the index case, e.g. by pooling samples 10-by-10 and re-testing individual samples from positive batches. Genome sequencing performed on all positive samples would help retrace transmission chains and help identify superspreaders and infection hotspots, whether in the current situation of epidemiological remission or if cases number increases.
We recommend that the testing capacity of Switzerland, currently around 20’000 tests per day and based exclusively on RT-PCR, be preemptively increased by: i) passing agreements with current providers for standing orders of RT-PCR tests throughout the year; ii) further exploration and validation of alternative methods, i.e. isothermal tests and NGS, the sensitivity of which may still be slightly below that of RT-PCR but stands to be improved; iii) integration of SARS-CoV-2 testing in multi-virus detection kits, including point-of-care diagnostics kits; iv) securing an activatable viral test production pipeline on Swiss ground, mobilizing Swiss-based companies engaged in the production of the necessary supplies, reagents and equipment.
5) What indicators should we use to decide on imposing additional measures in a situation where case numbers increase again? For example, are the daily number of newly confirmed cases or the number of hospitalizations good indicators? Can we use trend change (e.g. change from fluctuating case numbers to steadily increasing case numbers) as an indicator to switch from containment to mitigation 1? And can we use the absence of a trend change after a certain period of time as an indicator to switch from mitigation 1 to mitigation 2?
The daily number of new confirmed cases, hospitalizations, deaths, virological tests and ICU occupancy continue to be highly relevant indicators to monitor and evaluate the status of the epidemic, and are required for example to calculate the effective reproductive number, Re. With low numbers the estimate of Re comes with wide confidence intervals that include a Re of 1. This implies that there is uncertainty on whether the epidemic is growing (Re > 1) or not. Moreover, the closer Re is to 1, the longer the time series needs to be to detect whether Re is statistically different from 1. Hence, at low case numbers further indicators become relevant (see also answers to question 1), including:
- Traceability of contact tracing (i.e. what fraction of cases can be linked to other cases). Higher traceability indicates better coverage of all transmission chains by contact tracing.
- Time delay between the onset of symptoms in an index case and the isolation of the index case as well as his/her contacts. A shorter delay implies a larger positive effect in preventing further transmission of the disease by the index case and his/her contacts.
- Real time information on source of infection of index cases, allowing the identification of hotspots of transmission (events and locations).
- Changes of seropositivity in sentinel populations (such as patients admitted to hospitals, healthcare workers, residents in retirement homes, schools). Comparisons of the prevalence of seropositivity and the cumulative number of confirmed cases allow assessment of the extent to which the number of confirmed cases underestimates the extent of the epidemic.
- An important indicator of the need for hospital and ICU capacity is the condition of the individuals that have recently been infected by SARS-CoV-2. These factors include gender, age, BMI, diabetes, hypertension, co-morbidities and co-medications. Collecting this information in all confirmed and hospitalized patients would allow the prediction of the upcoming demand on health infrastructure, and could serve as a basis for deciding on the introduction of additional measures.
6) What can we say about the effectiveness of different measures? (benefits quantified as effects on Re; of course, we have to consider that measures can interact, in the sense that the effect of a measure can depend on whether or not another measure is in place). It is also useful to know if one cannot make conclusive statements about the benefits of individual measures.
A first important and timely question pertains to the effectiveness of face masks. Of interest are both whether masks prevent an infected wearer to spread the infection to others and whether they prevent a non-infected wearer from getting infected. This is a topic of broad interest to the general population also because of different policies used in neighboring countries. A new systematic review and meta-analysis commissioned by the WHO and published on June 1, 2020, addresses the effects of face masks on SARS-CoV-2, SARS-and MERS-transmission (source). The use of face masks was found to result in a large reduction in the risk of infection of up to 80%. The authors stressed that confounding factors cannot be ruled out and that the strength of the certainty of the evidence was thus low. Nevertheless, this study supports the notion that a person wearing a mask does not only reduce the risk of infecting others but also reduces the risk of becoming infected, in line with the earlier recommendations made by the Swiss National COVID-19 Science Task Force.
A second important question is about the effectiveness of general, population-wide measures on the curbing of the epidemic. In general, the quantification of effectiveness is difficult, because multiple measures are often implemented at once and the effect of one measure may depend on what other measures have been implemented. A recent report by the European Center for Disease Control and Prevention (ECDC) (link) came to the conclusion that the effectiveness of each individual type of control measure is unknown. To overcome the issue that measuring the effectiveness is statistically challenging, the ECDC conducted a survey among experts. Based on this expert assessment, the effectiveness of enforced stay-at-home orders was deemed highest, followed by the cancellation of mass gathering events which had a slightly lower overall effectiveness.
Despite this general challenge in separating different effects, there are a number of studies that attempt at estimating the effectiveness of different measures. Askitas et al. (2020, source) studied new COVID-19 cases and mobility in 134 countries, with the aim to disentangle the effects of measures to control COVID-19: international travel controls, public transport closure, cancellation of public events, restrictions on gatherings, school closure, workplace closure, stay-at-home requirements, and restrictions on internal movement. Results indicate that cancelling public events and restrictions on gatherings reduced the growth in the number of cases after about 14 days. School closures and workplace closures also reduced the growth in cases, after more than 14 days. Stay-at-home orders (curfews) were issued in times of rapid growth of cases and reduced growth somewhat, but not below the level of countries without stay-at-home orders. International travel controls, public transport closure, and restrictions on domestic movements had little effect on the number of COVID-19 cases. Bonardi et al. (2020, source) studied lockdown policies in 184 countries and found that measures targeting mobility inside countries reduced the growth in COVID-19 cases more effectively than measures targeting mobility across countries.
7) What can we say about the costs of different measures?
(costs in terms of economic consequences, consequences for the society and individuals, or health consequences)
The costs to society can roughly be divided into three categories. The first is due to the international environment. As a small open economy, Switzerland is well integrated into international supply chains and produces goods and services for foreign markets. Sectors that are linked to the international economy are hit (almost) independently of an increase in cases and/or domestic policy measures to contain the pandemic. According to KOF estimates, more than half of all economic costs are linked to the international environment (source and source). The second relates to behavioural changes and the real costs associated with people being sick and unable to work. Even in the absence of any policy measures, the fear of illness will have a considerable economic cost. A recent study on South Korea, a country without lockdown and only localised impact of COVID-19, shows that the impact on the economy is still considerable (source). The third category of costs to society is directly triggered by policy measures.
All three and especially the last two are connected and difficult to disentangle. In fact, the economic costs could indeed be higher without appropriate measures in place. Therefore, not all costs can or should be attributed to active containment policies. However, as long as the virus is relatively well contained, measures at the individual level (TTIQ and other targeted measures, including social distancing, hygiene and mask wearing) are almost always less costly (to society and the economy) than restrictions at the population level.
Different sectors or parts of society are affected differently by restrictions. Labour intensive occupations and professions that require close contact are more affected. Looking at short-time work statistics, the hotel, restaurant, arts, entertainment and leisure sectors are most strongly affected.
8) Which measures are suitable to be used for a regional approach?
For example, measures that can easily be circumvented by the population by driving to a different region (e.g. shop closures) are less straight-forward to implement regionally.
The strategy of surveillance-response aims at, as illustrated above, detecting spatially confined increases in case numbers and transmission early, which in turn will allow response measures to be tailored to the given outbreak situation, for example, outbreaks in school settings, religious and cultural events, funerals, nursing homes. Outbreaks across national borders will follow similar patterns and can be handled with the respective cross-border arrangements in place from 15 June onwards (Policy Brief in preparation). The measures tailored to a given cluster of transmission / outbreak will entail the whole range of measures discussed above, such as reinforcing hygiene, distance rules, selective mask wearing to temporary closures of businesses or schools. We stress that the strategy of surveillance-response aims to prevent any major regional or national lockdown.
9) Which innovations or events have the potential to be game-changing?
For example vaccination and effective treatment would be major opportunities. Are there also major risks?
Vaccination: Having effective and safe vaccines available would change the situation radically and could resolve the COVID-19 crisis. In a first phase, we will have incomplete information about the effectiveness and safety of newly developed vaccines; also, we expect that vaccines might initially only be available in limited quantities. During this first phase, specific immunisation strategies could be considered, such as directly targeting only those at highest risks (e.g. the frail elderly, patients in nursing homes, etc.). Even such a targeted strategy is however unlikely before 2021, given the current state of clinical development of the most advanced vaccine candidates and the decreasing case numbers that currently prevent performing efficacy trials in many areas including Europe.
Pharmacological interventions: Sars-CoV-2 leads to mild or moderate disease in approximately 80% of cases, but to serious or very serious disease in 20%. The predictors of serious COVID-19 are well described (see also Policy Brief on predictors). The introduction of highly effective antiviral or antiinflammatory treatments that improve prognosis considerably, or the development of highly effective drugs that prevent COVID-19 in people who have been exposed and have early or no symptoms have the potential to be game-changing. Unfortunately, no such pharmacologic interventions are available at present, or likely to be available in the near future. The taskforce is monitoring developments in this regard.
10) What health effects do individual measures have?
What are the effects of e.g. a lock-down on suicides, missed treatments for conditions other than COVID-19 etc? Are there also positive health effects, e.g. increased levels of physical activity by the population?
The measures imposed in mid March 2020 had both positive as well as negative health effects. On the positive side, the measures were associated with a two-fold increase in bicycle riding (source) and increased visits of outdoor recreation areas. Also, there was a marked reduction in injuries from traffic accidents. On the negative side, there was a reduction in the capacity for outpatient care of patients with chronic conditions (e.g. diabetes). While this could partially be compensated for by transferring care to hospitals, there is a clear risk that missed medical checks lead to increased risks of health deteriorations. This effect should be considered in future situations of increased case numbers.
To address these health effects in future scenarios with increased case numbers, we make the following recommendations:
- In future situations where COVID-19 cases increase, major efforts have to be made to maintain non-COVID-19 medicine in hospitals and private practices.
- Infection prevention and control measures have to be reinforced in the health care setting (hospitals and private practices) to ensure maintained patient care.
- In the presence of infection prevention and control measures, health care settings do not pose an elevated infection risk. Even if case numbers increase in future scenarios, we therefore recommend maintaining medical treatment of all patients and not restrict care to patients with urgent health conditions.
- Hospitals with a high case load may reduce the number of outpatients to decrease general workload and reduce the program for elective surgery. The latter increases ICU capacities for COVID-19 patients and non-COVID-19 patients in emergency situations.
- It is important to inform patients that healthcare settings do not pose elevated risks of contracting SARS-CoV2 to prevent that people do not help.
A number of studies also addressed impacts on the COVID-19 situation on mental health. The psychological reactions to the crisis likely depended on nation-specific socio-economic and healthcare factors and nation-specific lockdown measures. The survey of the Swiss Corona Stress Study ( source) in over 10’000 individuals revealed that psychological reactions to the lockdown in Switzerland were diverse: While 24% of the participants reported no change in stress levels, 50% of the participants reported an increase and 26% a decrease in stress levels during lockdown. The changes in stress levels were highly correlated with changes in depressive symptoms. The prevalence of moderately severe or severe depressive symptoms raised from 3.4% (before the corona crisis) to 9.1% (during lockdown), of which 20% did not complain about depressive symptoms before the crisis. Prevalence of daily suicidal thoughts raised from 0.8% (before the corona crisis) to 1.5% (during lockdown).
The study also revealed which lock-down related factors were most strongly related with distress and relief, respectively. Distress was caused by changes at work or schools, burden of childcare, loneliness and not being able to spend time with others. Decreases in stress were associated with relief at work and more time for relaxation.
To address these mental health effects in future scenarios with increased case numbers, we make the following recommendations:
- Systematically monitor mental health consequences.
- Inform the public about potential mental health consequences and the importance to seek professional help if needed.
Date of request: 26/05/2020
Date of response: 08/06/2020
In response to request from: Hans Matter FOPH
Comment on planned updates: –
Expert groups and individuals involved: All expert groups and the advisory board contributed to this document.
Contact persons:
Martin Ackermann (martin.ackermann@env.ethz.ch), Sebastian Bonhoeffer (seb@env.ethz.ch), Matthias Egger (Matthias.egger@snf.ch)